Bioanalytical and CMC Services for small molecules, biologics and
advanced therapies for the life science industry
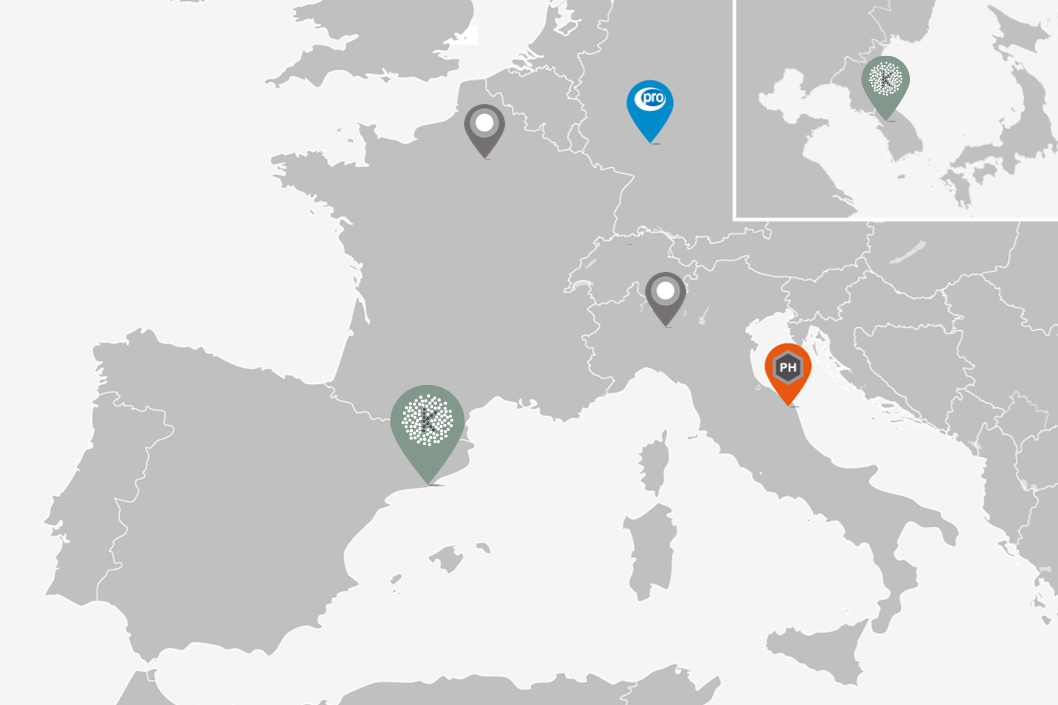
The Kymos Group is an analytical CRO with three European laboratories devoted to providing bioanalytical and CMC services for the life science industry throughout the entire product life cycle: from early research and development to manufacturing and commercialization. We are GLP- and GMP-certified, have been successfully inspected by the EMA and the FDA and are experts in analytical and quality control work with small molecules, biologics and oligonucleotides. Kymos provides services to clients worldwide in the pharmaceutical, biotechnology, veterinary, fine chemistry, cosmetic and nutraceutical industries.
Testimonials

“We are currently developing multiple new biosimilar projects and consider Kymos one of the best CROs in Europe for drug release and testing.”

“We were positively impressed when Kymos reacted to our needs by investing in their stability capabilities, and now, two years down the line, with projects in full-swing, I can safely say that we made the right choice. In Kymos we have found a reliable, committed, trustworthy and professional partner.”

“Back in 2017 there weren’t many analytical CROs able to carry out Franz cell assays and Kymos’ pioneering work was pivotal for the registration of our product.”

“Kymos offered superior historical expertise by being exposed to so many different clients, pharmacological compounds, and therapeutic areas. They addressed and anticipated very diverse problems.”

“I have had a long and fruitful experience with Kymos and have a huge interest in their participation owing to their reliability.”

“Prolytic is our analytical partner throughout the entire drug development process. Such a long and complex process requires a laboratory you can trust. Professional yet flexible, Prolytic accompanies us through the preclinical and clinical phases.”

“Establishing robust miRNA methods for valuable results is tricky. The team at Prolytic have been a key collaboration partner. With their 30 years of experience in bioanalytics and broad know-how in different techniques, study types, and substance groups, we were able to quickly develop a reproducible and reliable analysis method for miRNAs.”
Latest News
Stability Testing: What’s to come in 2024
Stability testing is an essential part of drug development as it ensures the efficacy and safety of the medicinal product throughout its shelf life. It allows the evaluation of APIs and drug product stability under (...) Read more
Impurity Profiling: Characterization of unknown impurities in pharmaceuticals
Characterization of unknown impurities found in pharmaceutical substances and pharmaceutical products is a vital part of the regulatory approval process. According to ICH guidelines, if the estimation indicates an impurity content higher than 0.1%, it (...) Read more