BIOEQUIVALENCE STUDIES
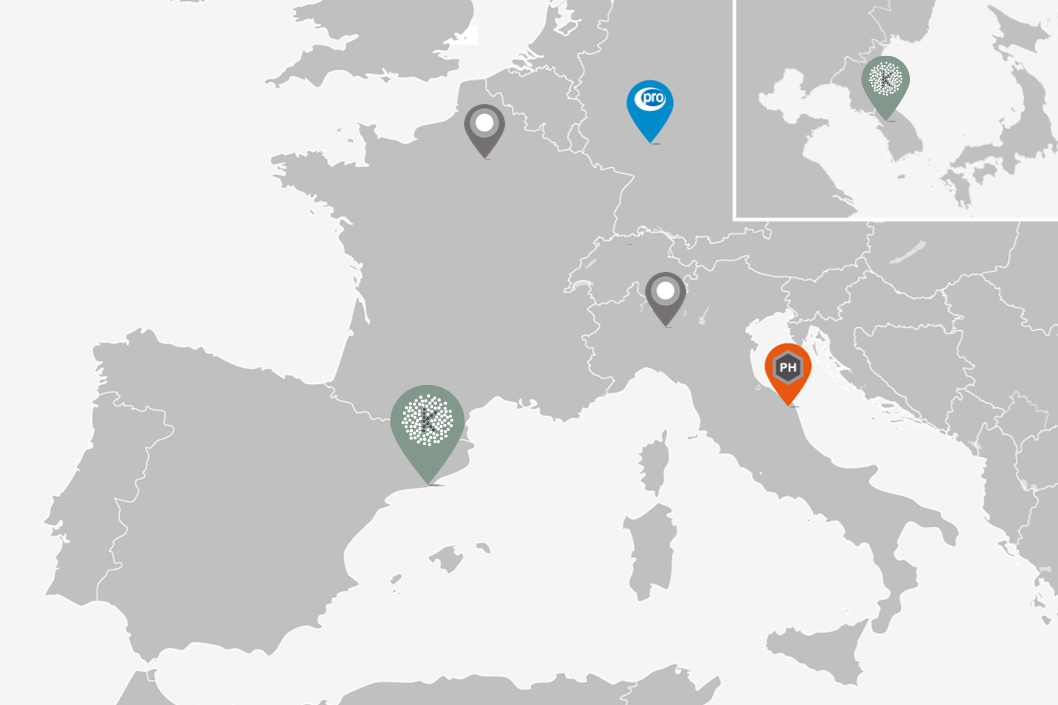
Our bioanalysis experts are experienced in the comprehensive management of bioequivalence studies, offering end-to-end service from IMPD review to final reporting. We work in alliance with reputed European clinical and preclinical centers following EMA and FDA guidelines. High-quality standards, narrow timelines and competitive prices are key elements for our delivery. We are fully coordinated with our alliance partners to provide One Stop Shop services for bioequivalence studies in the following areas:

BIOEQUIVALENCE STUDIES MANAGEMENT | BIOEQUIVALENCE STUDIES REPORTING |
|---|---|
|
|
BIOANALYSIS FOR BIOEQUIVALENCE STUDIES | SUPPORT ACTIVITIES TO BIOEQUIVALENCE STUDIES |
|
|
Our experts for bioequivalence studies can transfer existing bioanalytical methods and have a long list of validated methods offering the Clients the opportunity of directly analyzing their samples thereby saving time and money. If one is not available, we can develop and validate the method free of charge in a timely manner.